In this lecture, we begin our discussion of Optical Biosensors. We start off by showing the general properties of optical biosensors besides the SPR. Then, we shift our attention to the improvements made to the SPR Sensor's performance, but also limitations. Before moving onto the structure of the optical biosensor, we had to get an overview of the Thin Film Interference and its role as creating a reflection along with a refractive index for the light traveling through. We focus on a type optical biosensor called the Reflection Interference Spectroscopy (RIFS). The equation to measure the phase is seen compared to the structure of the biosensor. The output provides a means for measuring the reflection as a wavelength. As the distance of the interference layer increases, we see the period of the curve of the output begin to shift to the right. We then view a model of a biosensor that uses the RIFS system and the equipment setup.
My research group is focused on the application of sub-wavelength optical phenomena and fabrication methods to the development of novel devices and instrumentation for the life sciences. The group is highly interdisciplinary, with expertise in the areas of microfabrication, nanotechnology, computer simulation, instrumentation, molecular biology, and cell biology. In particular, we are working on biosensors based upon photonic crystal concepts that can either be built from low-cost flexible plastic materials, or integrated with semiconductor-based active devices, such as light sources and photodetectors, for high performance integrated detection systems.
Using a combination of micrometer-scale and nanometer-scale fabrication tools, we are devising novel methods and materials for producing electro-optic devices with nanometer-scale features that can be scaled for low-cost manufacturing. Many of our techniques are geared for compatibility with flexible plastic materials, leading to applications such as low cost disposable sensors, wearable sensors, flexible electronics, and flexible displays. Because our structures manipulate light at a scale that is smaller than an optical wavelength, we rely on computer simulation tools such as Rigorous Coupled Wave Analysis (RCWA) and Finite Difference Time Doman (FDTD) to model, design, and understand optical phenomena within photonic crystals and related devices.
In addition to fabricating devices, our group is also focused on the design, prototyping, and testing of biosensor instrumentation for high sensitivity, portability, and resolution. Advanced instruments enable high resolution imaging of biochemical and cellular interactions with the ability to monitor images of biochemical interactions as a function of time. Using the sensors and instrumentation, we are exploring new applications for optical biosensor technology including protein microarrays, biosensor/mass spectrometry systems, and microfluidics-based assays using nanoliter quantities of reagents. The methods and systems developed in the laboratory are applied in the fields of life science research, drug discovery, diagnostic testing, and environmental monitoring. -From Professor Cunningham's Faculty Profile
Researchers should cite this work as follows:
 1. Optical Biosensors Lecture 18 …
0
00:00/00:00
1. Optical Biosensors Lecture 18 …
0
00:00/00:00 2. Optical Biosensors Besides SPR
181.70462759549287
00:00/00:00
2. Optical Biosensors Besides SPR
181.70462759549287
00:00/00:00 3. Optical Biosensors Besides SPR
443.913034233468
00:00/00:00
3. Optical Biosensors Besides SPR
443.913034233468
00:00/00:00 4. Optical Biosensor Examples
566.01701041888157
00:00/00:00
4. Optical Biosensor Examples
566.01701041888157
00:00/00:00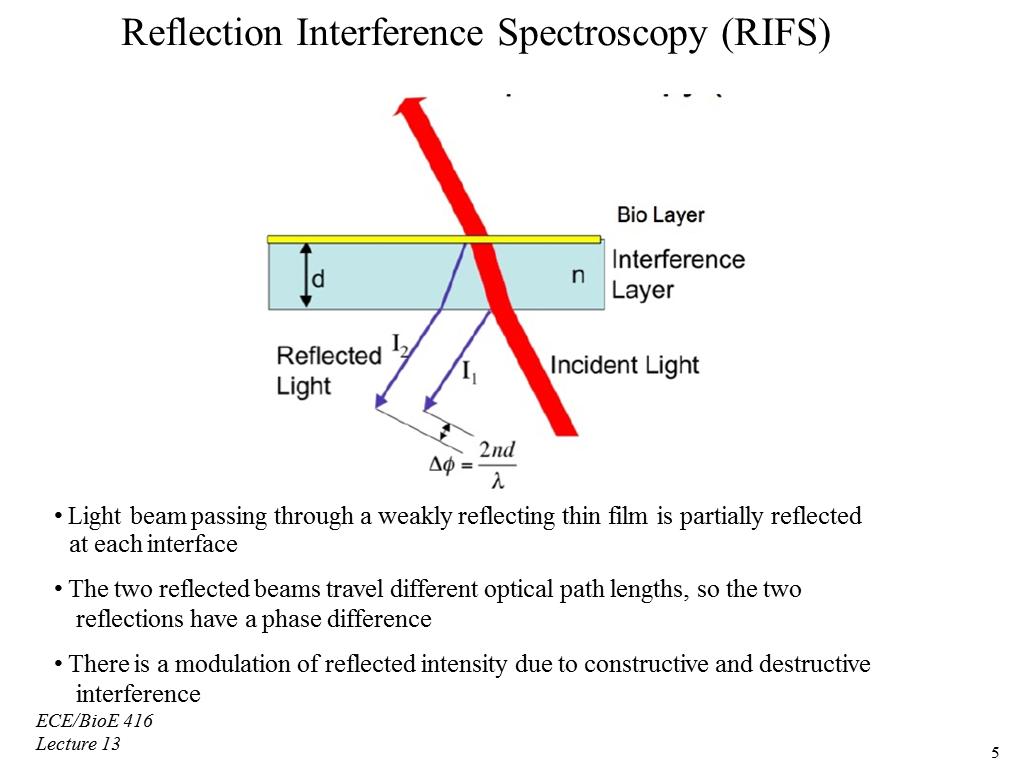 5. Reflection Interference Spectr…
1043.2522854510667
00:00/00:00
5. Reflection Interference Spectr…
1043.2522854510667
00:00/00:00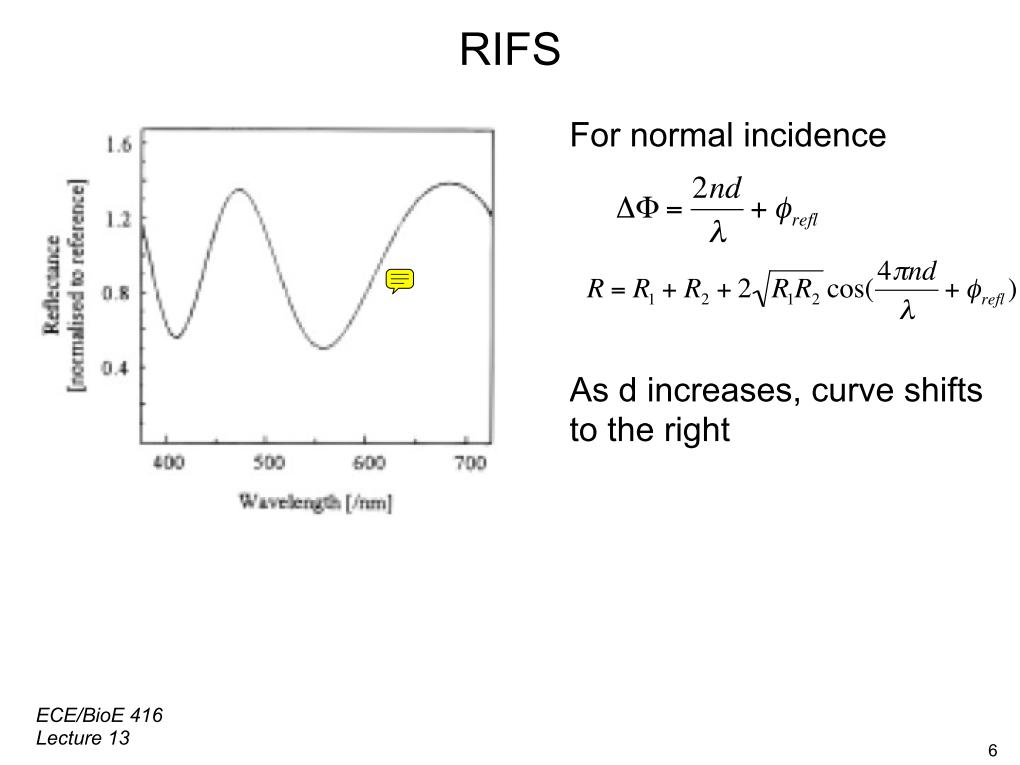 6. RIFS
1382.0682543057624
00:00/00:00
6. RIFS
1382.0682543057624
00:00/00:00 7. RIFS
1612.1443759302574
00:00/00:00
7. RIFS
1612.1443759302574
00:00/00:00 8. RIFS Data (Piehler Paper)
1671.770784605571
00:00/00:00
8. RIFS Data (Piehler Paper)
1671.770784605571
00:00/00:00