[Illinois] ECE 416 Avidin-Biotin and Surface Functionalization I
[Illinois] ECE 416 Lecture 29: Avidin-Biotin & Surface Functionalization I
-
 1. Lecture 20: The Avidin-Biotin …
0
00:00/00:00
1. Lecture 20: The Avidin-Biotin …
0
00:00/00:00 -
 2. Reminder: Our Surface Function…
33.21533923303835
00:00/00:00
2. Reminder: Our Surface Function…
33.21533923303835
00:00/00:00 -
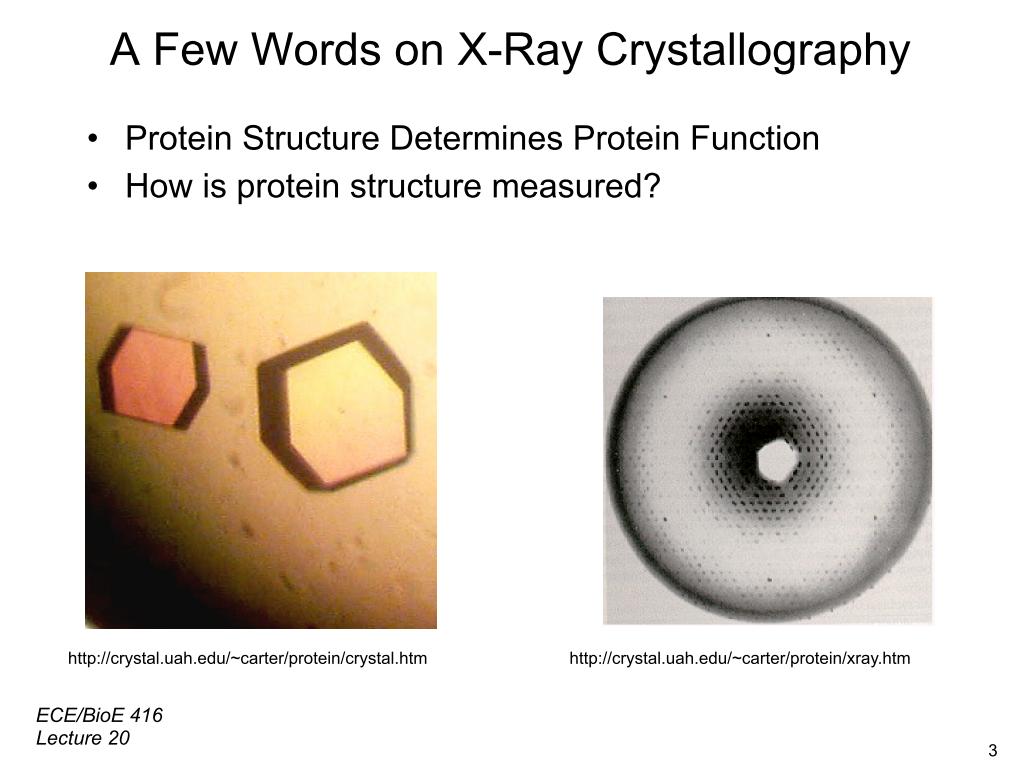 3. A Few Words on X-Ray Crystallo…
77.974062004579878
00:00/00:00
3. A Few Words on X-Ray Crystallo…
77.974062004579878
00:00/00:00 -
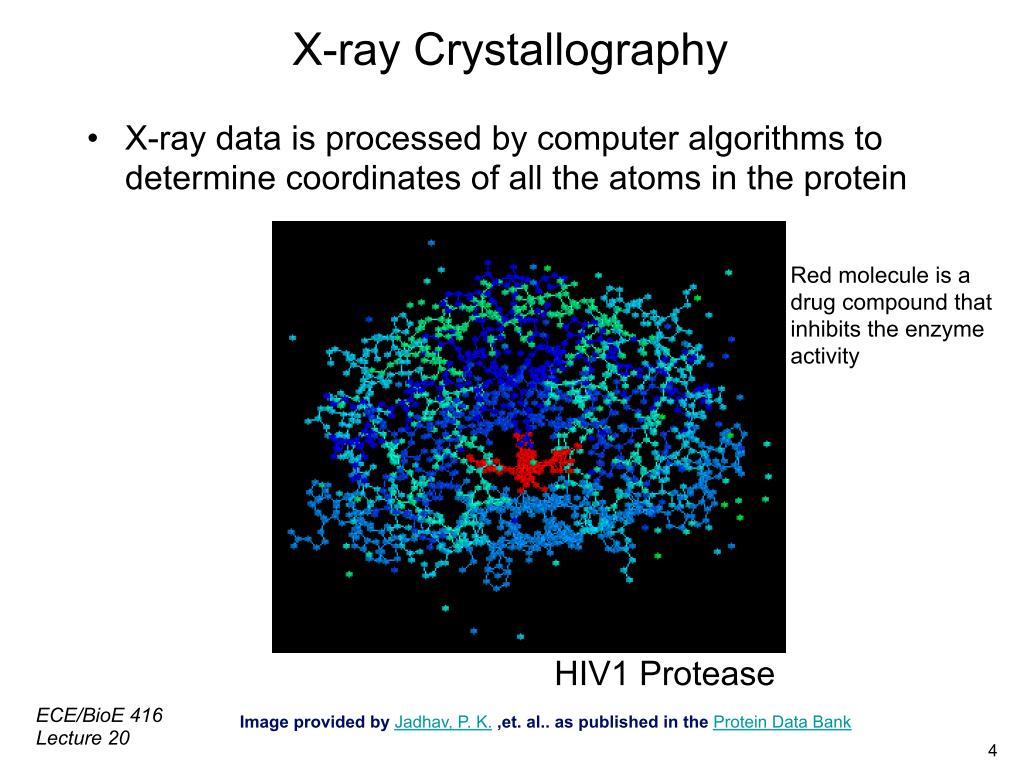 4. Xray Crystallography
93.717632552404439
00:00/00:00
4. Xray Crystallography
93.717632552404439
00:00/00:00 -
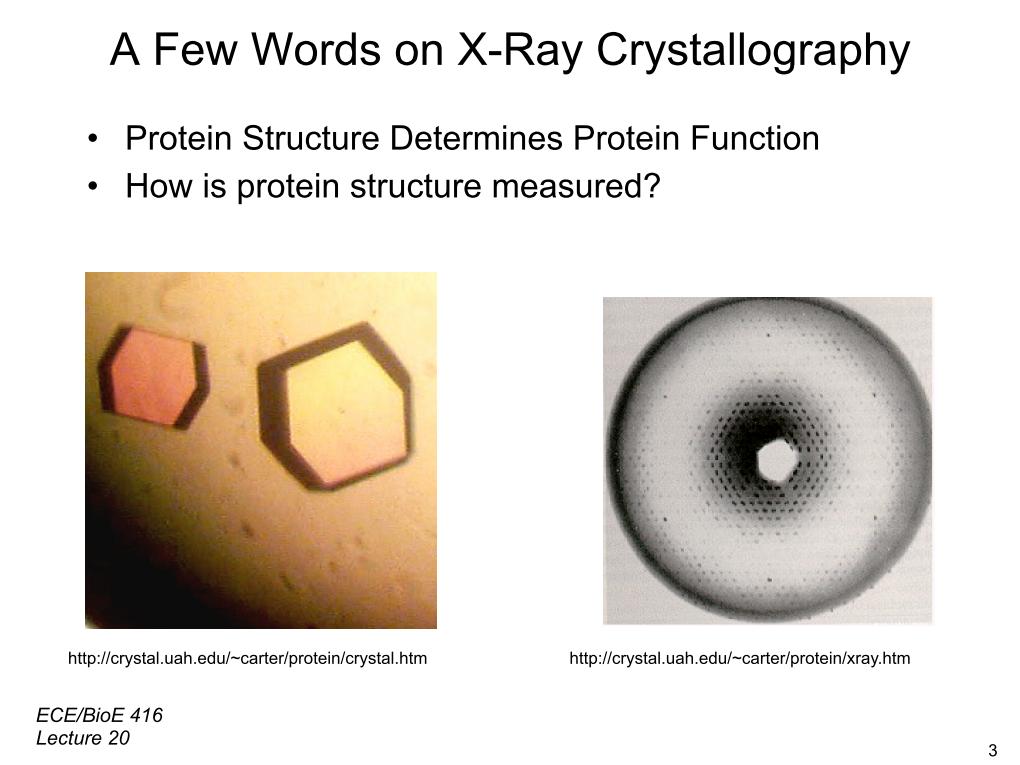 5. A Few Words on X-Ray Crystallo…
128.55183195349656
00:00/00:00
5. A Few Words on X-Ray Crystallo…
128.55183195349656
00:00/00:00 -
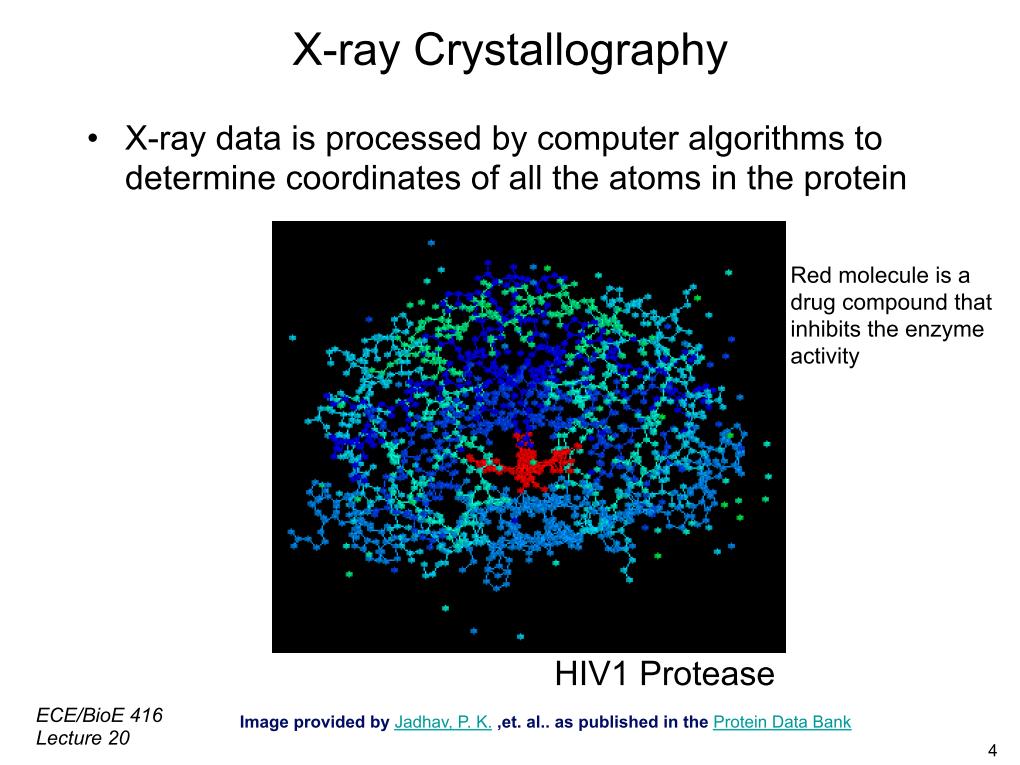 6. Xray Crystallography
307.557468733486
00:00/00:00
6. Xray Crystallography
307.557468733486
00:00/00:00 -
 7. Avidin
496.10841994010923
00:00/00:00
7. Avidin
496.10841994010923
00:00/00:00 -
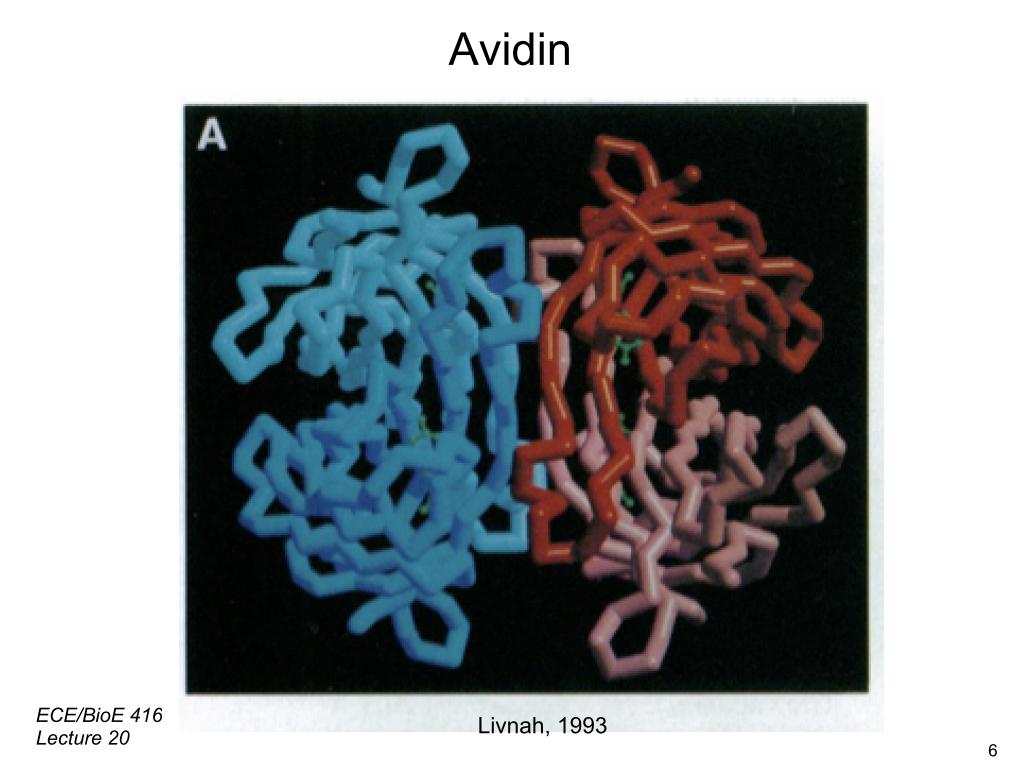 8. Avidin
580.03280782103218
00:00/00:00
8. Avidin
580.03280782103218
00:00/00:00 -
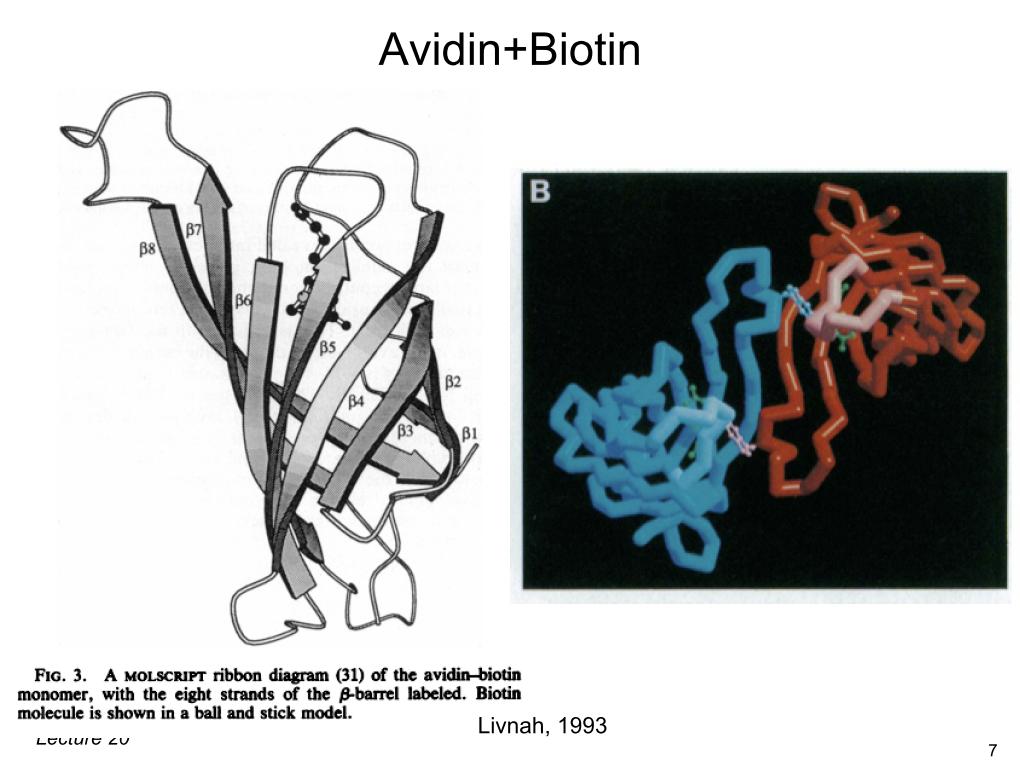 9. Avidin+Biotin
666.80839351770294
00:00/00:00
9. Avidin+Biotin
666.80839351770294
00:00/00:00 -
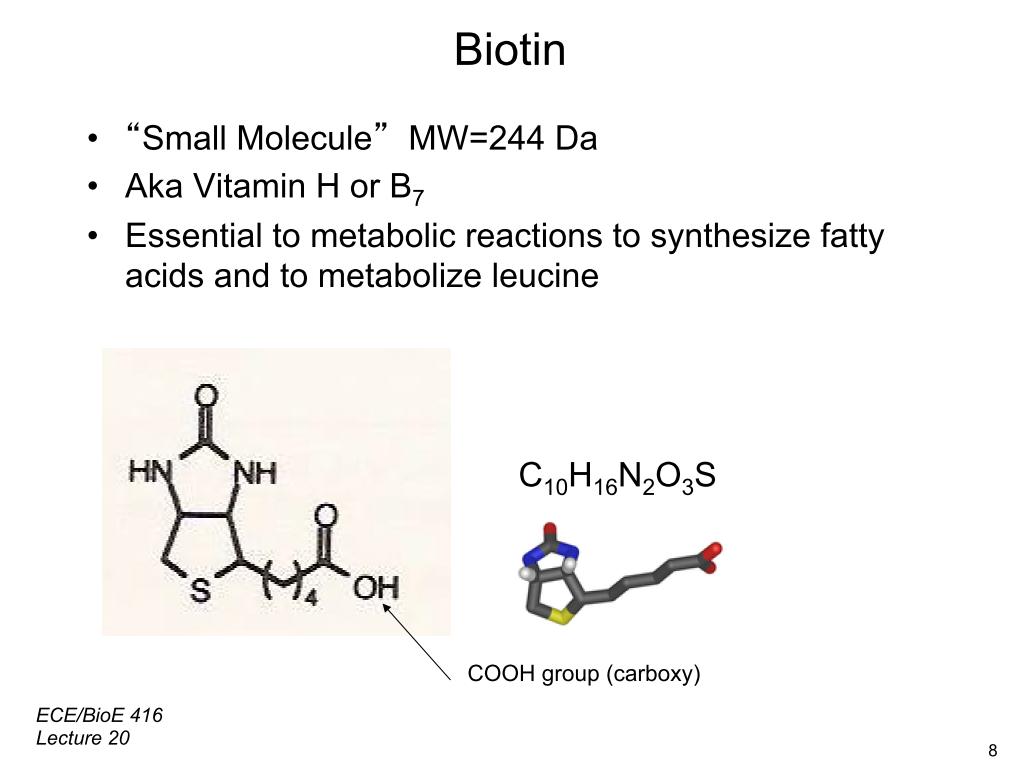 10. Biotin
716.02254711995784
00:00/00:00
10. Biotin
716.02254711995784
00:00/00:00 -
 11. Avidin-Biotin: Nature's Superg…
825.7316804650344
00:00/00:00
11. Avidin-Biotin: Nature's Superg…
825.7316804650344
00:00/00:00 -
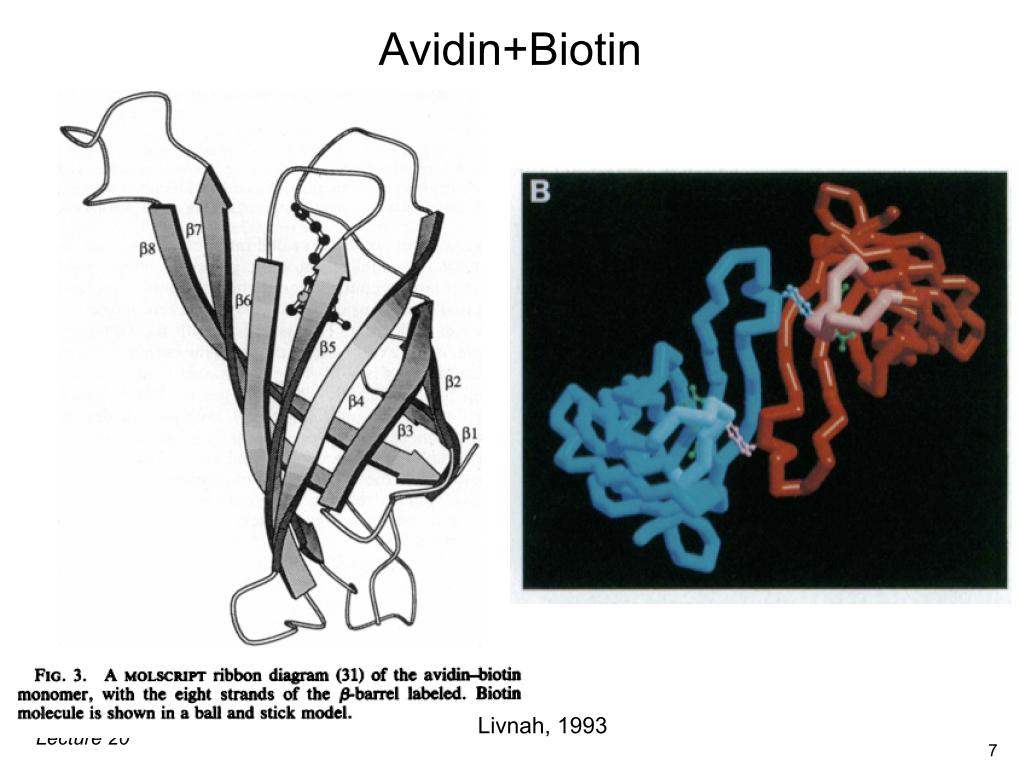 12. Avidin+Biotin
926.88722036286777
00:00/00:00
12. Avidin+Biotin
926.88722036286777
00:00/00:00 -
 13. Avidin-Biotin: Nature's Superg…
1088.9096353707944
00:00/00:00
13. Avidin-Biotin: Nature's Superg…
1088.9096353707944
00:00/00:00 -
 14. Livnah Paper on Avidin-Biotin
1107.7523339792144
00:00/00:00
14. Livnah Paper on Avidin-Biotin
1107.7523339792144
00:00/00:00 -
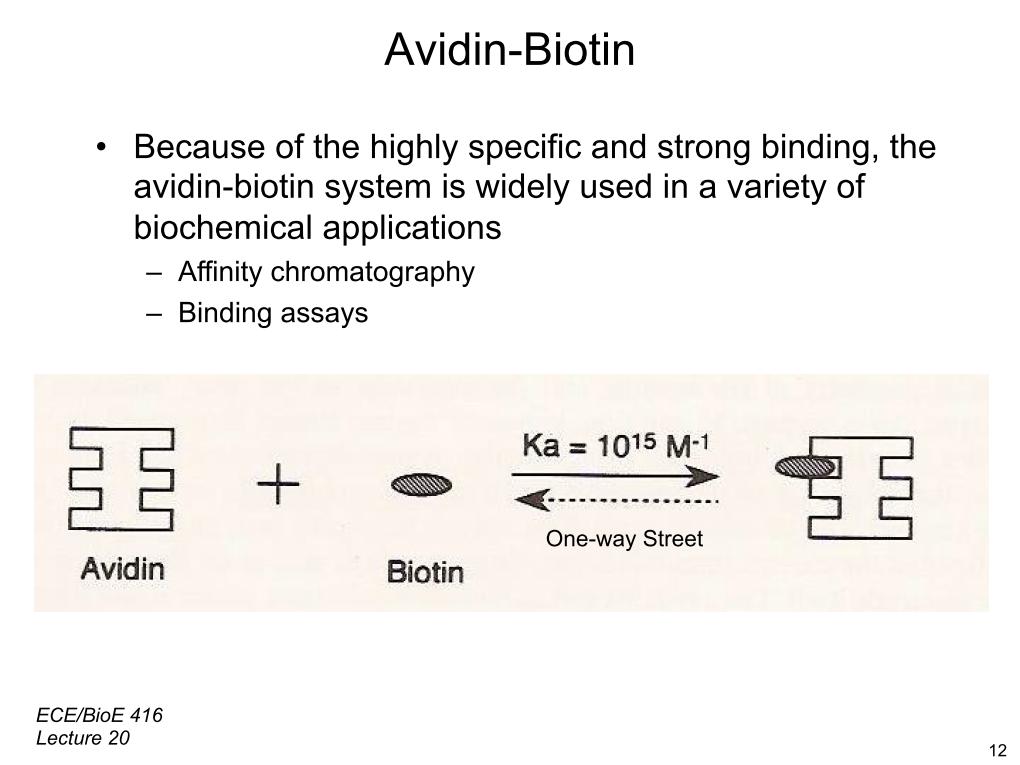 15. Avidin-Biotin
1258.2459926017264
00:00/00:00
15. Avidin-Biotin
1258.2459926017264
00:00/00:00 -
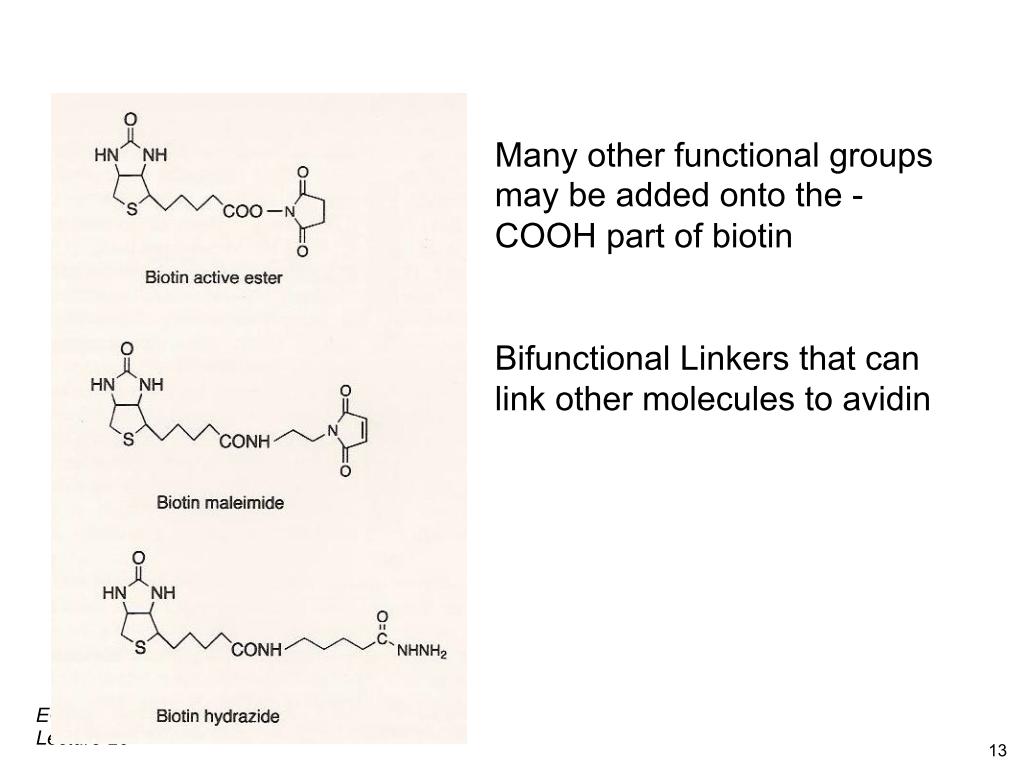 16. Biotinylation
1304.2370530209619
00:00/00:00
16. Biotinylation
1304.2370530209619
00:00/00:00 -
 17. Biotinylation
1346.7570900123305
00:00/00:00
17. Biotinylation
1346.7570900123305
00:00/00:00 -
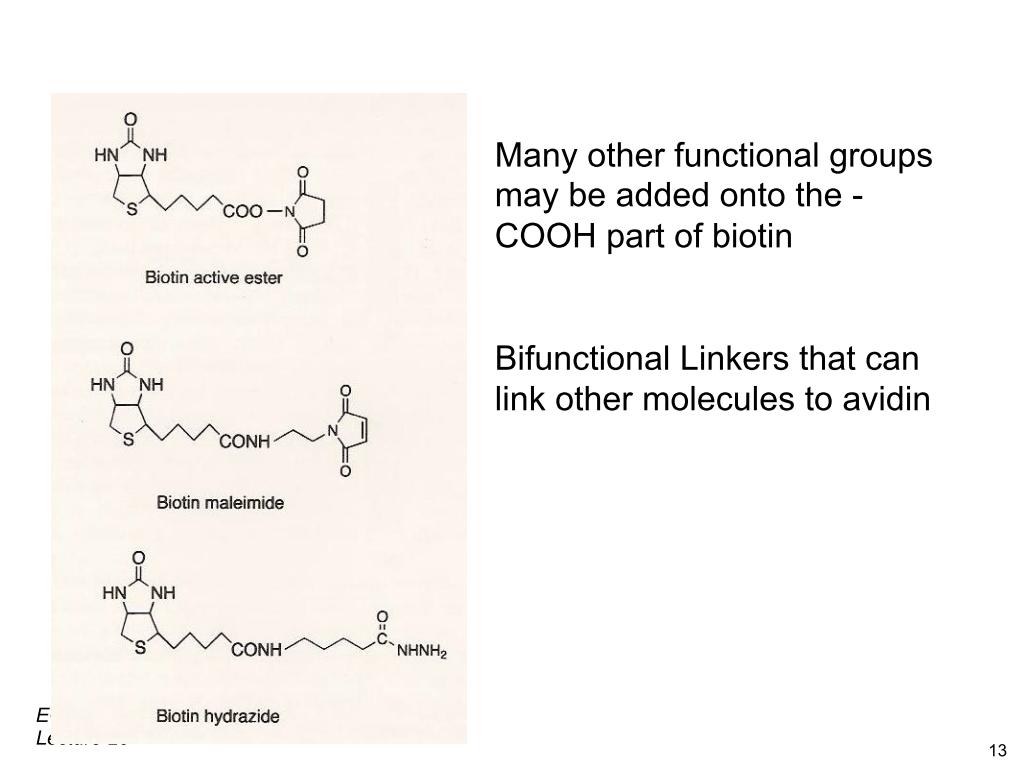 18. Biotinylation
1409.7313722036288
00:00/00:00
18. Biotinylation
1409.7313722036288
00:00/00:00 -
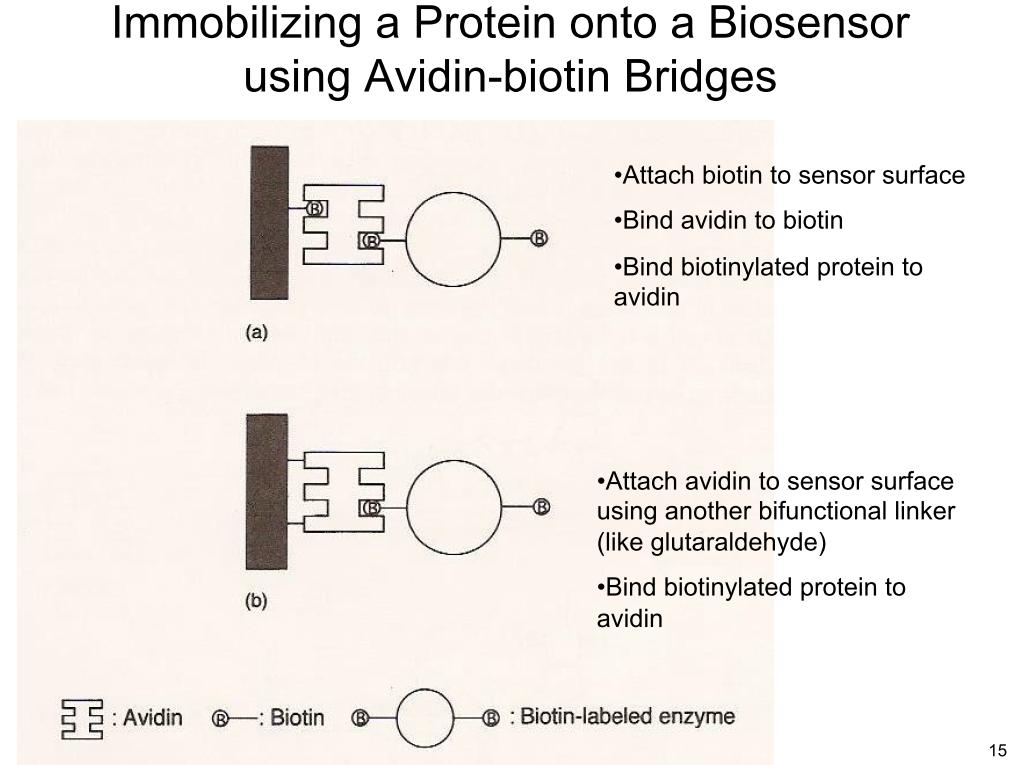 19. Immobilizing a Protein
1451.4359617840005
00:00/00:00
19. Immobilizing a Protein
1451.4359617840005
00:00/00:00 -
 20. Uses of Avidin-Biotin System
1556.2581469085785
00:00/00:00
20. Uses of Avidin-Biotin System
1556.2581469085785
00:00/00:00 -
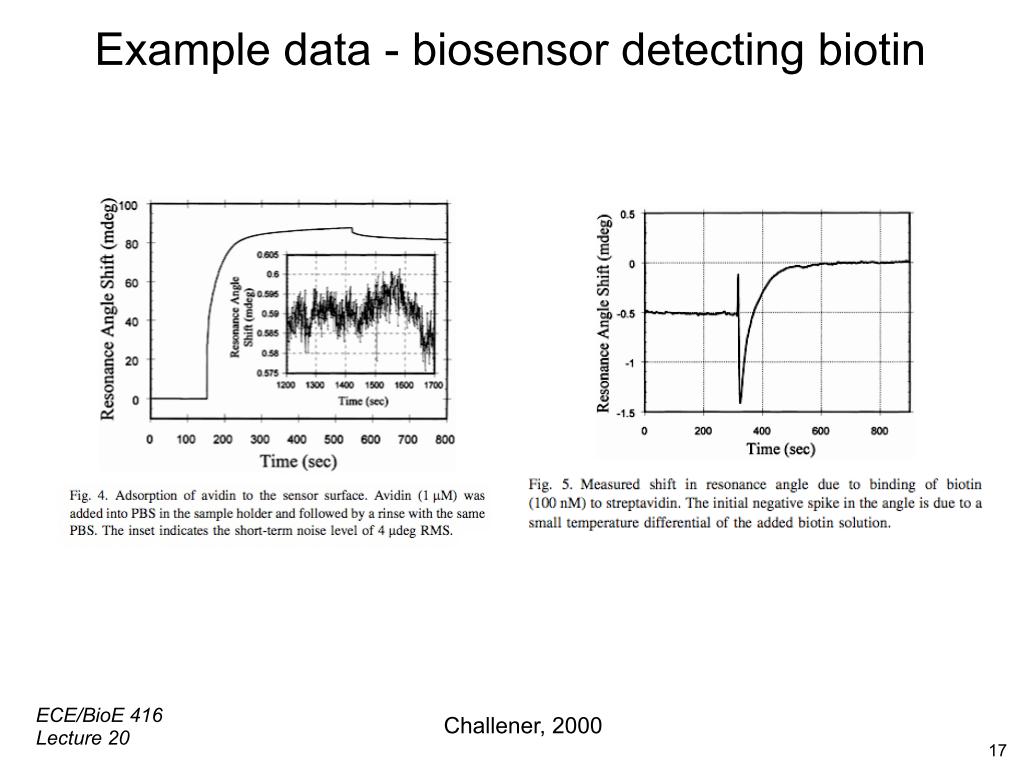 21. Example Data
1594.3154394926898
00:00/00:00
21. Example Data
1594.3154394926898
00:00/00:00 -
 22. Streptavidin (SA)
1656.9178263167166
00:00/00:00
22. Streptavidin (SA)
1656.9178263167166
00:00/00:00 -
 23. Avidin-Biotin System
1749.6437378897303
00:00/00:00
23. Avidin-Biotin System
1749.6437378897303
00:00/00:00 -
 24. Surface Functionalization Tech…
1929.2692002818389
00:00/00:00
24. Surface Functionalization Tech…
1929.2692002818389
00:00/00:00 -
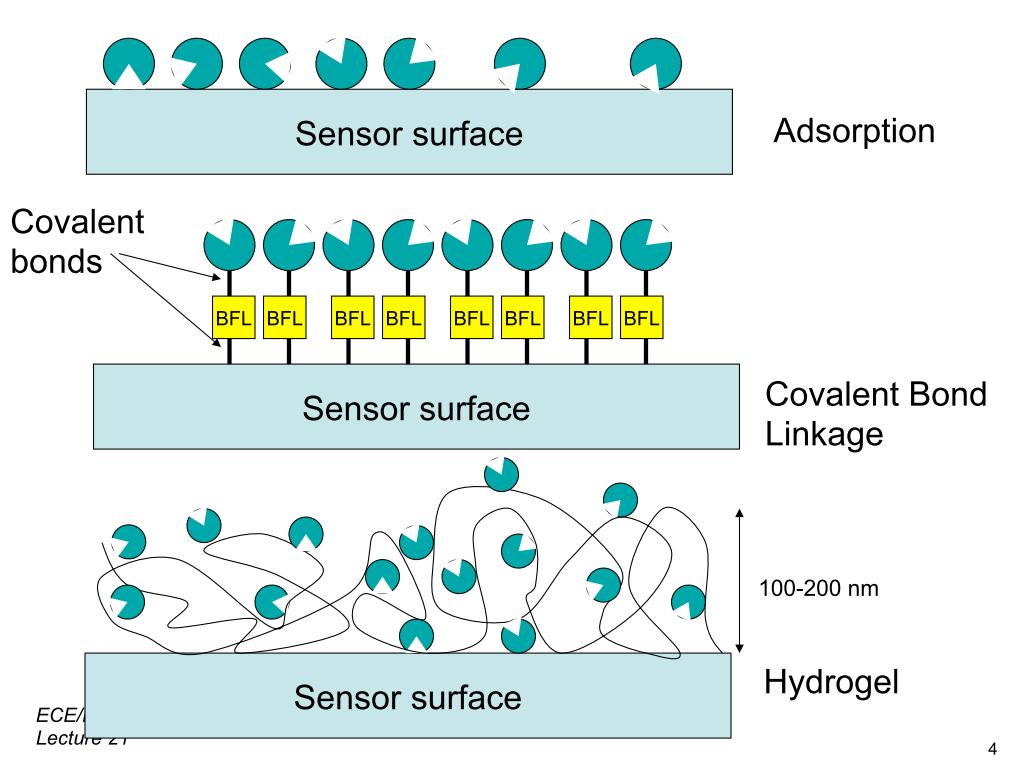 25. Surface Functionalization Tech…
2055.2177646644354
00:00/00:00
25. Surface Functionalization Tech…
2055.2177646644354
00:00/00:00 -
 26. Adsorption
2128.1092566496391
00:00/00:00
26. Adsorption
2128.1092566496391
00:00/00:00 -
 27. Driving Forces for Adsorption
2200.5048881451471
00:00/00:00
27. Driving Forces for Adsorption
2200.5048881451471
00:00/00:00 -
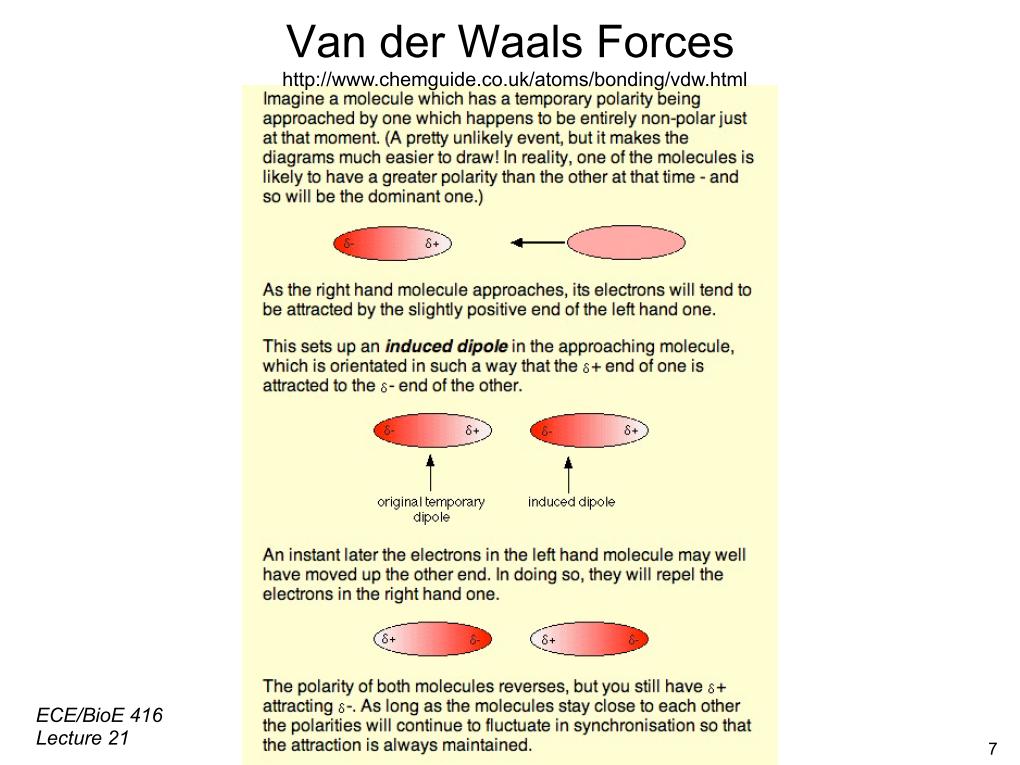 28. Van Der Waals Forces
2288.8920204333276
00:00/00:00
28. Van Der Waals Forces
2288.8920204333276
00:00/00:00 -
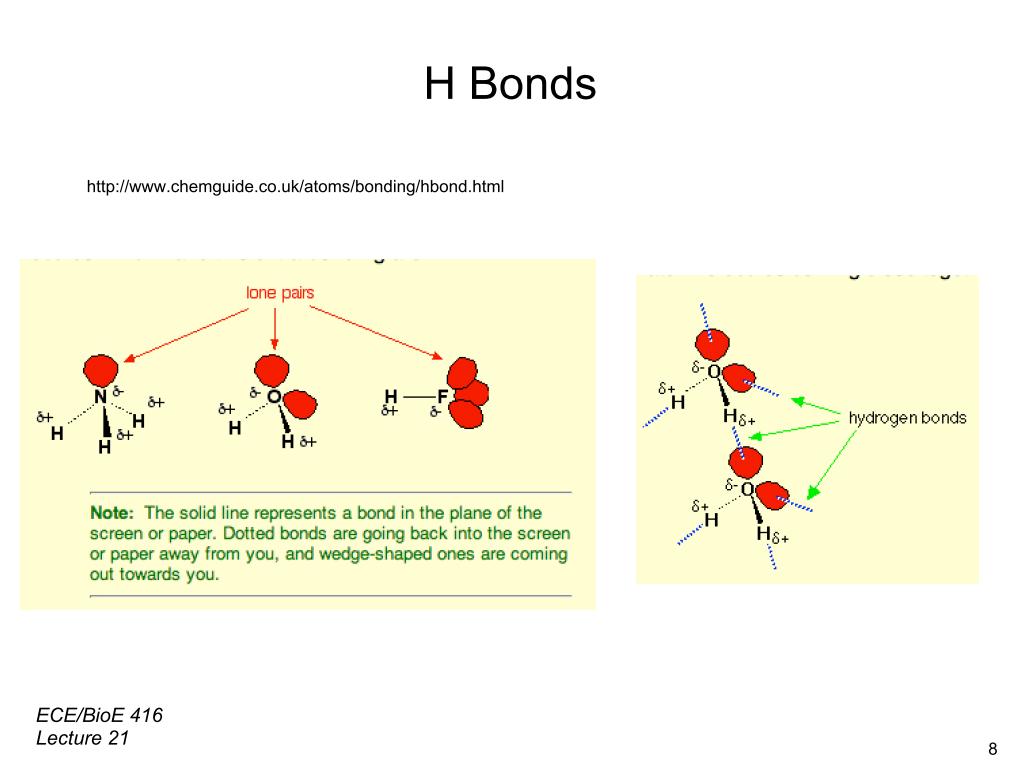 29. H Bonds
2387.6922230051086
00:00/00:00
29. H Bonds
2387.6922230051086
00:00/00:00 -
 30. The Problems with Adsorption
2475.5834948035936
00:00/00:00
30. The Problems with Adsorption
2475.5834948035936
00:00/00:00 -
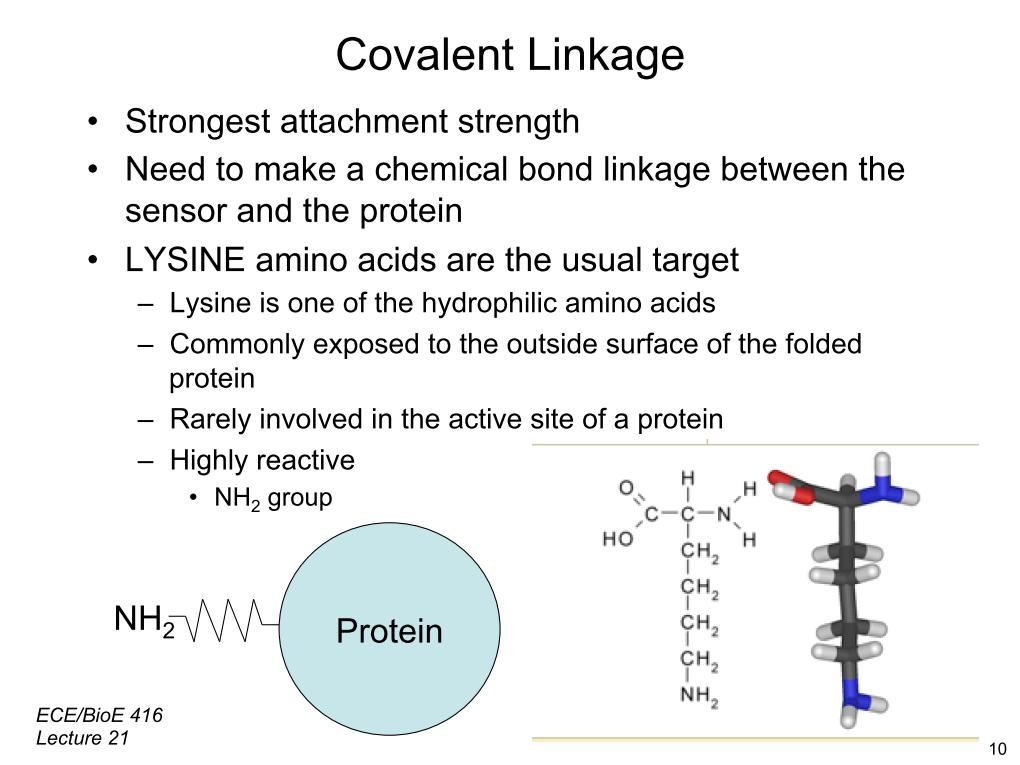 31. Covalent Linkage
2523.55799718161
00:00/00:00
31. Covalent Linkage
2523.55799718161
00:00/00:00 -
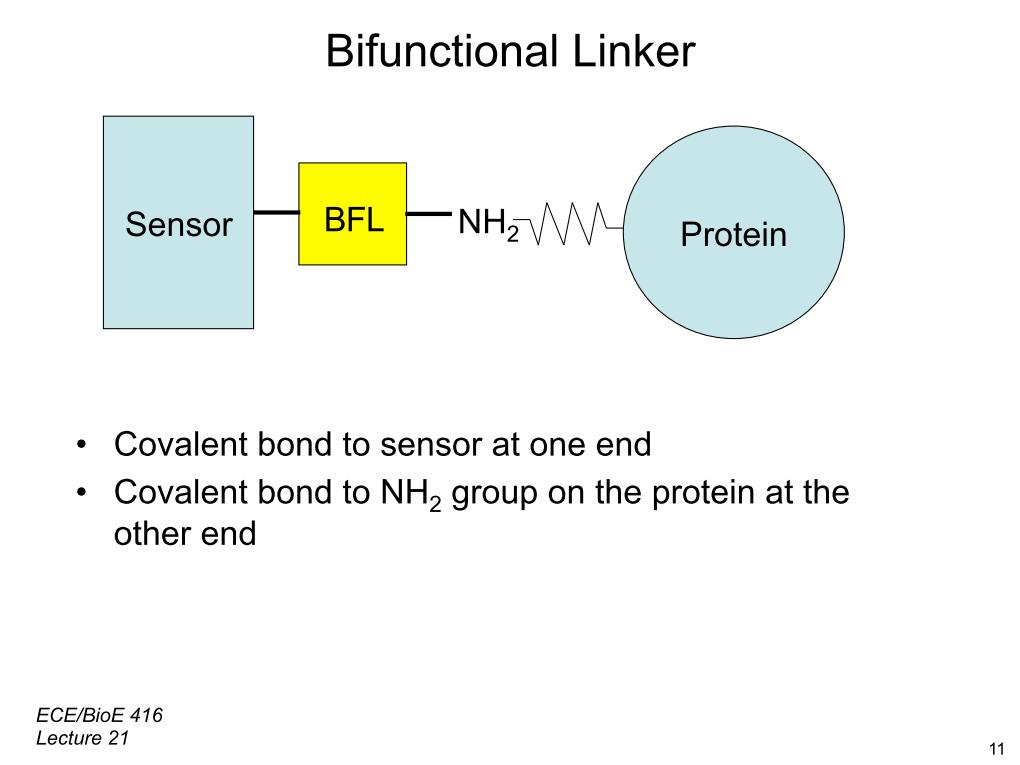 32. Bifunctional Linker
2602.5237801655803
00:00/00:00
32. Bifunctional Linker
2602.5237801655803
00:00/00:00 -
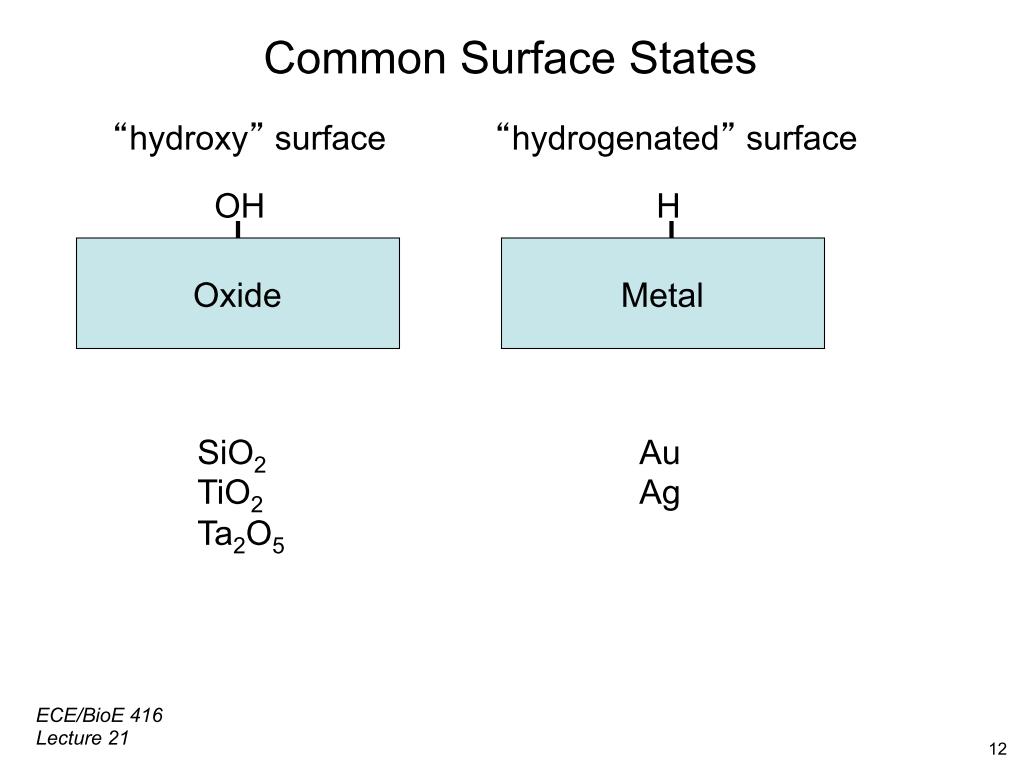 33. Common Surface States
2646.4074335036112
00:00/00:00
33. Common Surface States
2646.4074335036112
00:00/00:00 -
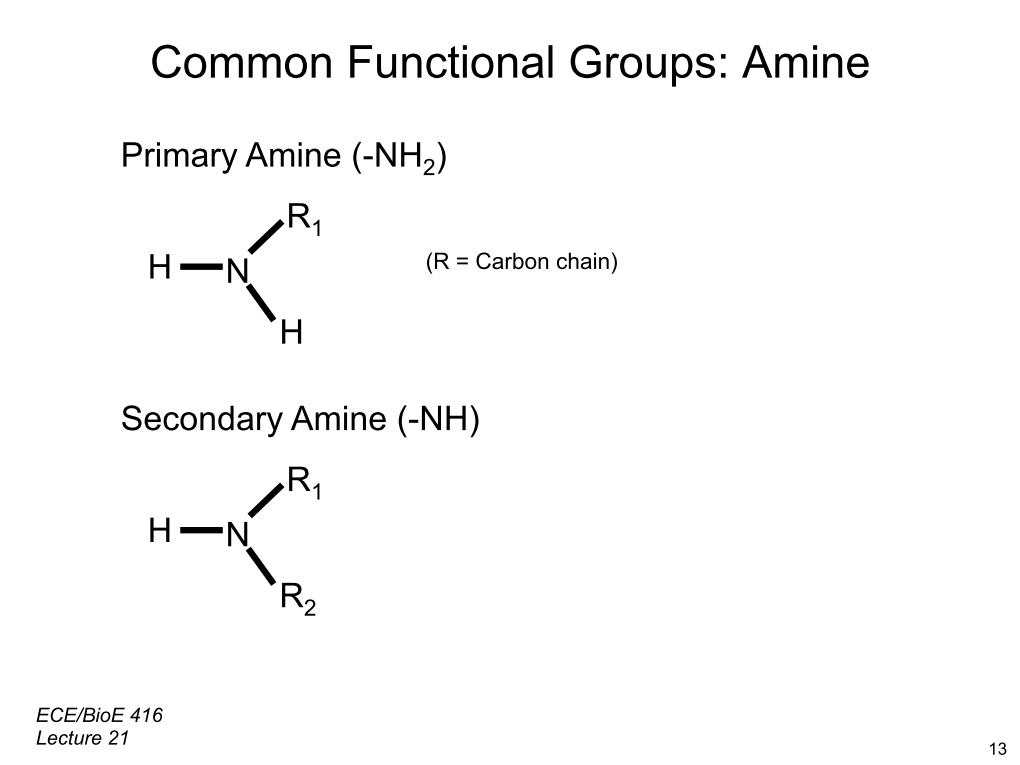 34. Common Funtional Groups: Amine
2711.3651576536904
00:00/00:00
34. Common Funtional Groups: Amine
2711.3651576536904
00:00/00:00 -
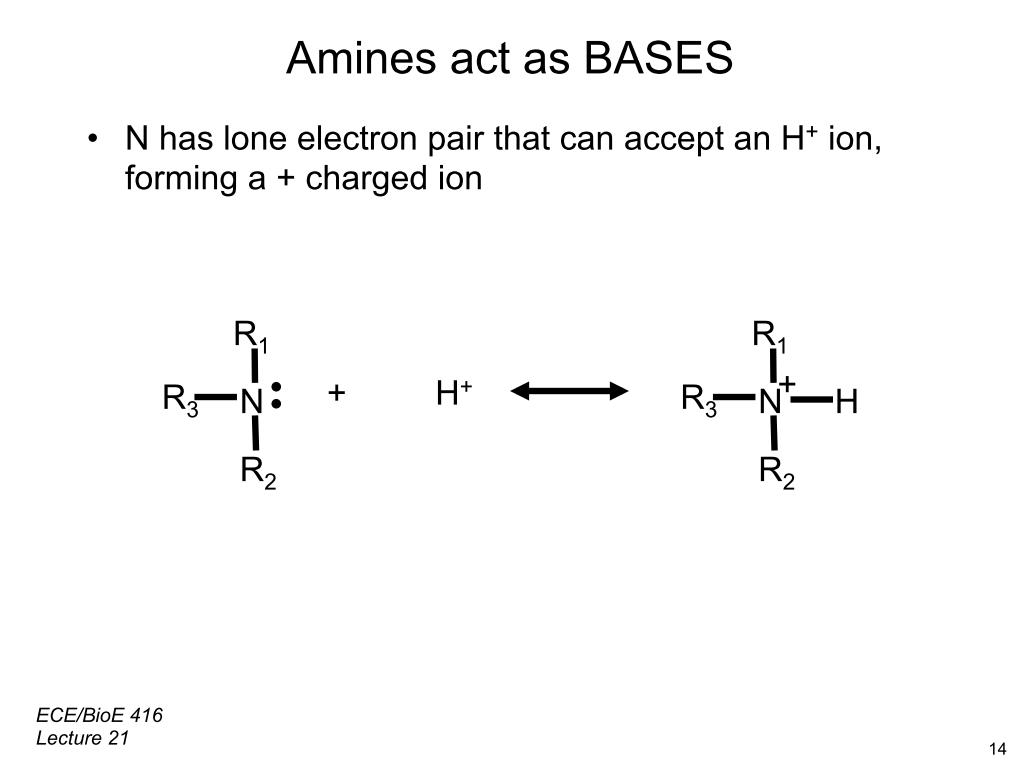 35. Amine acts at BASES
2758.2239739298925
00:00/00:00
35. Amine acts at BASES
2758.2239739298925
00:00/00:00